|
There you can find the metals, semi-conductor (s), non-metal (s), inert noble gas (ses), Halogens, Lanthanoides. Please note that the elements do not show their natural relation towards each other as in the Periodic system. Visualize trends, 3D orbitals, isotopes, and mix compounds. The unity for atomic mass is gram per mol. Interactive periodic table showing names, electrons, and oxidation states. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. The lightest chemical element is Hydrogen and the heaviest is Hassium. Element Radon (Rn), Group 18, Atomic Number 86, p-block, Mass 222. 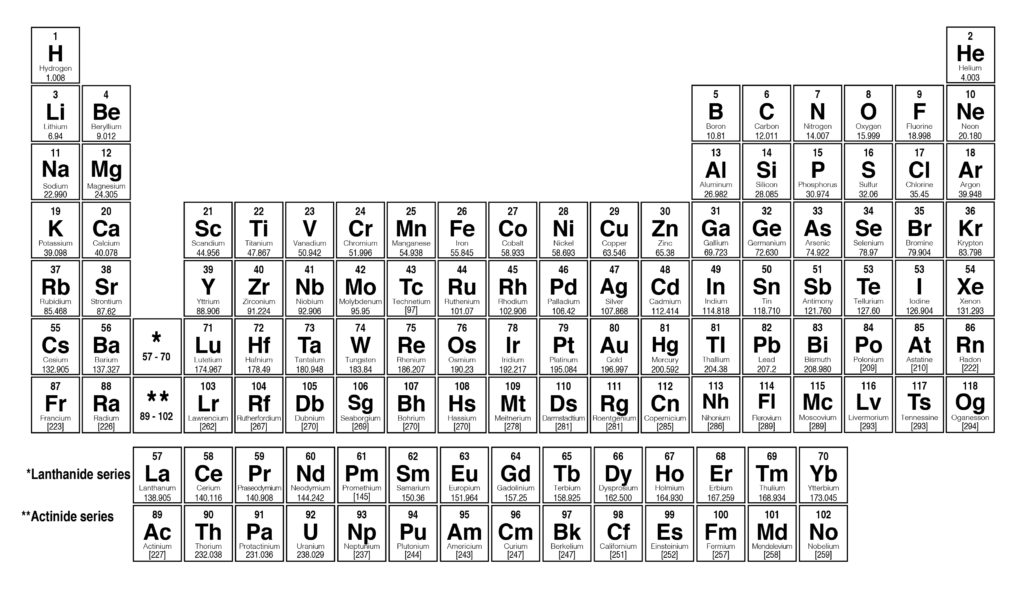
Atoms are so small, however, that even 500 atoms are too small to see or measure by most common techniques. Periodic Table of Videos Created by video journalist Brady Haran working with chemists at The University of Nottingham. Sheets of printer paper are packaged in reams of 500, a seemingly large number. For example, cans of soda come in a six-pack, eggs are sold by the dozen (12), and pencils often come in a gross (12 dozen, or 144). Many familiar items are sold in numerical quantities with distinct names. No single carbon atom has a mass of 12.01 amu, but. This is the average atomic mass of carbon. The unit that provides this link is the mole (mol), from the Latin moles, meaning “pile” or “heap.” On the periodic table the mass of carbon is reported as 12.01 amu. What is the molecular mass of acetaminophen (C8H9NO2). The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6.022 × 10 23 carbon atomsis therefore 12.011 g/mol: Table 2.4.1 2.4. The molecular weight of a compound is the sum of the atomic masses of all atoms in the molecule. You can also use this molarity calculator to find the mass concentration or molar mass. Substitute the known values to calculate the molarity: molarity 5 / (1.2 × 36.46) 0.114 mol/l 0.114 M. 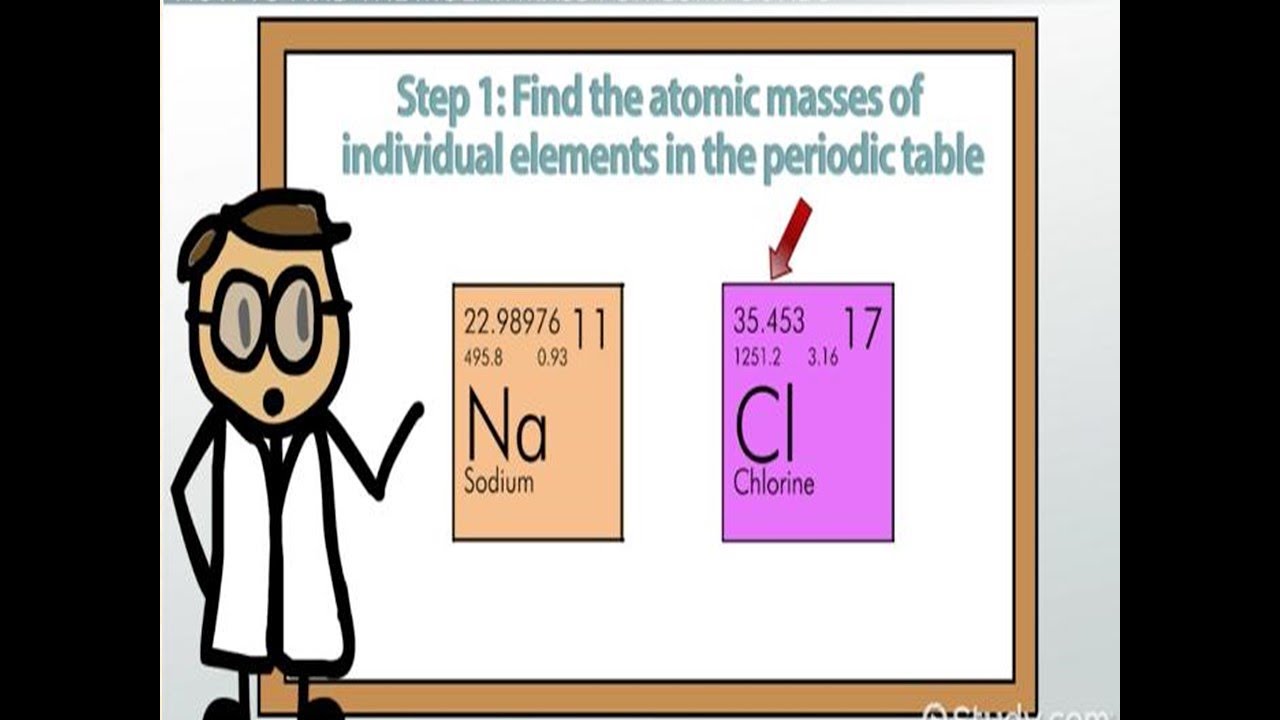
As mass / volume molarity × molar mass, then mass / (volume × molar mass) molarity. To analyze the transformations that occur between individual atoms or molecules in a chemical reaction, it is therefore essential for chemists to know how many atoms or molecules are contained in a measurable quantity in the laboratory-a given mass of sample. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. Convert the expressions above to obtain a molarity formula. In the laboratory, for example, the masses of compounds and elements used by chemists typically range from milligrams to grams, while in industry, chemicals are bought and sold in kilograms and tons. Because the masses of individual atoms are so minuscule (on the order of 10 −23 g/atom), chemists do not measure the mass of individual atoms or molecules. The problem for Dalton and other early chemists was to discover the quantitative relationship between the number of atoms in a chemical substance and its mass. The molar mass of a compound is equal to the sum of the molar masses of the. One atomic mass unit (u) is equal to 1/12 the mass of one atom of carbon-12. Molecular weight of compounds : from Acetaldehyde to calcium chloride Ammonium oxide, (NH4)2O, 52.0763 g/mol Ammonium persulfate, (NH4)2S2O8, 228.18 g/mol. the mass in grams of 1 mol of an element (the atomic mass on the periodic table). 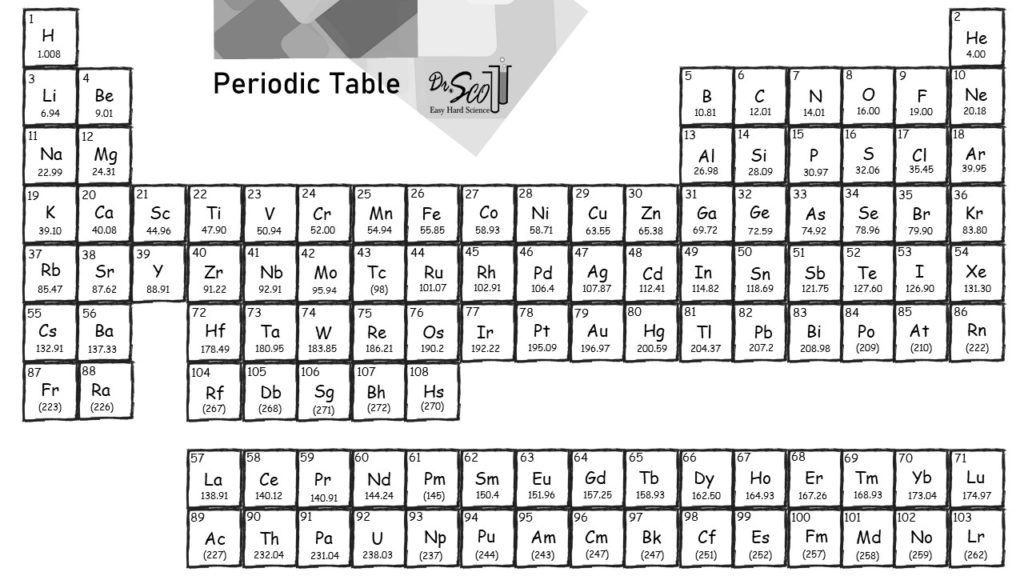
It also describes the law of multiple proportions, which states that the ratios of the masses of elements that form a series of compounds are small whole numbers. Molar mass is the mass (in atomic mass units) of one mole of a of a substance. Make sure you enter the molecule of crystallization at last (e.g. The conversion factors are 1 mole Cl2 over 70.90 grams Cl2, and 6.022 times 10^23 Cl2 molecules over 1 mole Cl2._3(PO_4)_2 = 310.177 \, amu \nonumber \]ĭalton’s theory that each chemical compound has a particular combination of atoms and that the ratios of the numbers of atoms of the elements present are usually small whole numbers. Prepare a concept map and use the proper conversion factor. Identify the "given" information and what the problem is asking you to "find."ġ mol Cl 2 = 70.90 g Cl 2, 1mol Cl 2 = 6.022 x 10 23 Cl 2 molecules
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
